Indonesia Yet Starting Covid-19 Vaccination for 6-11 Years Old Children
Indonesia has yet to begin the Covid-19 vaccination for children aged 6-11 despite the issuance on t...

Indonesia has yet to begin the Covid-19 vaccination for children aged 6-11 despite the issuance on t...

BPOM issues the emergency use authorization (EUA) for the Johnson & Johnson vaccine and CanSino.

COVID-19 Task Force spokesman Wiku said the Russian-made COVID-19 vaccine Sputnik V is designed to b...

BPOM issued an Emergency Use Authorization (EUA) for the Sputnik V Covid-19 Vaccine on Tuesday, Augu...
Indonesian drug monitoring agency officially issues the emergency use authorization for Pfizer-BioNt...

BPOM has issued an emergency use authorization (EUA) for the Covid-19 vaccine produced by the United...

The BPOM has officially issued an emergency use authorization (EUA) for Sinopharm vaccines.

Food and Drug Monitoring Agency (BPOM) granted an emergency use authorization for the Covid-19 vacci...

The Food and Drug Monitoring Agency (BPOM) officially issues an emergency use authorization (EUA) fo...
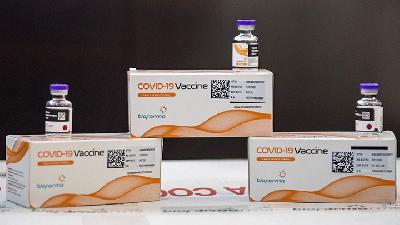
BPOM chairperson Penny Lukito said the emergency use authorization (EUA) had been issued for the COV...

BPOM Head Penny Lukito confirmed she was waiting for the results of the phase-3 clinical trial of t...